Ba Oh 2 Compound Name
 | |
 | |
| Identifiers | |
|---|---|
| CAS Number |
|
| 3D model (JSmol) |
|
| ChEBI |
|
| ChemSpider |
|
| ECHA InfoCard | 100.037.470 |
| EC Number |
|
| Gmelin Reference | 846955 |
| PubChem CID |
|
| RTECS number |
|
| UNII |
|
| CompTox Dashboard (EPA) |
|
| InChI
| |
| SMILES
| |
| Properties | |
| Chemical formula | Ba(OH)2 |
| Tooth mass | 171.34 g/mol (anhydrous) 189.355 g/mol (monohydrate) 315.46 chiliad/mol (octahydrate) |
| Advent | white solid |
| Density | three.743 thou/cm3 (monohydrate) two.xviii g/cm3 (octahydrate, sixteen °C) |
| Melting betoken | 78 °C (172 °F; 351 Chiliad) (octahydrate) 300 °C (monohydrate) 407 °C (anhydrous) |
| Boiling point | 780 °C (1,440 °F; ane,050 K) |
| Solubility in water | mass of BaO (not Ba(OH)ii): 1.67 chiliad/100 mL (0 °C) 3.89 g/100 mL (20 °C) 4.68 g/100 mL (25 °C) 5.59 g/100 mL (thirty °C) 8.22 grand/100 mL (40 °C) 11.7 m/100 mL (50 °C) 20.94 thou/100 mL (lx °C) 101.4 one thousand/100 mL (100 °C)[ citation needed ] |
| Solubility in other solvents | low |
| Basicity (p1000 b) | 0.xv (first OH–), 0.64 (second OH–)[ane] |
| Magnetic susceptibility (χ) | −53.ii·10−half dozen cm3/mol |
| Refractive index (n D) | 1.50 (octahydrate) |
| Structure | |
| Crystal construction | octahedral |
| Thermochemistry[2] | |
| Std enthalpy of | −944.vii kJ·mol−1 |
| Enthalpy of fusion (Δf H ⦵ fus) | 16 kJ·mol−one |
| Hazards | |
| GHS labelling: | |
| Pictograms |   |
| Signal discussion | Danger |
| Chance statements | H302, H314, H332, H412 |
| NFPA 704 (fire diamond) | iii 0 0 |
| Flash point | Non-combustible |
| Related compounds | |
| Other anions | Barium oxide Barium peroxide |
| Other cations | Calcium hydroxide Strontium hydroxide |
| Supplementary data page | |
| Barium hydroxide (data page) | |
| Except where otherwise noted, information are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). Infobox references | |
Barium hydroxide is a chemic chemical compound with the chemical formula Ba(OH)2. The monohydrate (x = 1), known as baryta or baryta-water, is one of the principal compounds of barium. This white granular monohydrate is the usual commercial course.
Preparation and structure [edit]
Barium hydroxide can be prepared past dissolving barium oxide (BaO) in water:
- BaO + H2O → Ba(OH)2
It crystallises as the octahydrate, which converts to the monohydrate upon heating in air. At 100 °C in a vacuum, the monohydrate will yield BaO and water.[three] The monohydrate adopts a layered structure (see movie in a higher place). The Batwo+ centers prefer a square anti-prismatic geometry. Each Ba2+ center is bound by two water ligands and 6 hydroxide ligands, which are respectively doubly and triply bridging to neighboring Ba2+ centre sites.[4] In the octahydrate, the individual Batwo+ centers are once again 8 coordinate but do non share ligands.[5]
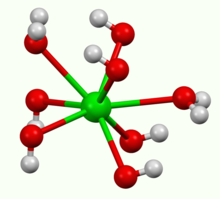
Coordination sphere about an individual barium ion in Ba(OH)2.H2O.
Uses [edit]
Industrially, barium hydroxide is used equally the forerunner to other barium compounds. The monohydrate is used to dehydrate and remove sulfate from various products.[6] This application exploits the very depression solubility of barium sulfate. This industrial application is also applied to laboratory uses.
Laboratory uses [edit]
Barium hydroxide is used in analytical chemistry for the titration of weak acids, specially organic acids. Its clear aqueous solution is guaranteed to exist free of carbonate, unlike those of sodium hydroxide and potassium hydroxide, as barium carbonate is insoluble in water. This allows the use of indicators such as phenolphthalein or thymolphthalein (with alkaline colour changes) without the risk of titration errors due to the presence of carbonate ions, which are much less basic.[7]
Barium hydroxide is occasionally used in organic synthesis as a strong base, for example for the hydrolysis of esters[eight] and nitriles,[9] [10] [xi] and as a base in aldol condensations.
In that location are several uses for barium hydroxide such equally to hydrolyse one of the two equivalent ester groups in dimethyl hendecanedioate.[12]
Barium hydroxide has besides been used, too, in the decarboxylation of amino acids liberating barium carbonate in the process.[13]
Information technology is also used in the training of cyclopentanone,[14] diacetone booze[xv] and D-gulonic γ-lactone.[16]
Reactions [edit]
Barium hydroxide decomposes to barium oxide when heated to 800 °C. Reaction with carbon dioxide gives barium carbonate. Its aqueous solution, being highly alkaline metal, undergoes neutralization reactions with acids due to it being a strong base. It is particularly useful on reactions that require the titrations of weak organic acids. Thus, it forms barium sulfate and barium phosphate with sulfuric and phosphoric acids, respectively. Reaction with hydrogen sulfide produces barium sulfide. Atmospheric precipitation of many insoluble, or less soluble barium salts, may result from double replacement reaction when a barium hydroxide aqueous solution is mixed with many solutions of other metal salts.[17]
Reactions of barium hydroxide with ammonium salts are strongly endothermic. The reaction of barium hydroxide octahydrate with ammonium chloride[18] [19] or[20] ammonium thiocyanate[20] [21] is frequently used as a classroom chemistry sit-in, producing temperatures cold enough to freeze water and plenty h2o to dissolve the resulting mixture.
Prophylactic [edit]
Barium hydroxide presents the same hazards such as skin irritation and burns likewise as centre impairment, just as the other strong bases and every bit other water-soluble barium compounds: information technology is corrosive and toxic.[ citation needed ]
Run into also [edit]
- Baralyme
References [edit]
- ^ "Sortierte Liste: pKb-Werte, nach Ordnungszahl sortiert. - Das Periodensystem online" (in High german).
- ^ Lide, David R., ed. (2009). CRC Handbook of Chemistry and Physics (90th ed.). Boca Raton, Florida: CRC Printing. ISBN978-1-4200-9084-0.
- ^ (1960). Gmelins Handbuch der anorganischen Chemie (8. Aufl.), Weinheim: Verlag Chemie, p. 289.
- ^ Kuske, P.; Engelen, B.; Henning, J.; Lutz, H.D.; Fuess, H.; Gregson, D. "Neutron diffraction study of Sr(OH)2(HiiO) and beta-Ba(OH)2*(H2O)" Zeitschrift für Kristallographie (1979-2010) 1988, vol. 183, p319-p325.
- ^ Manohar, H.; Ramaseshan, South. "The crystal structure of barium hydroxide octahydrate Ba (OH)2(H2O)eight" Zeitschrift für Kristallographie, Kristallgeometrie, Kristallphysik, Kristallchemie 1964. vol. 119, p357-p374
- ^ Robert Kresse, Ulrich Baudis, Paul Jäger, H. Hermann Riechers, Heinz Wagner, Jochen Winkler, Hans Uwe Wolf, "Barium and Barium Compounds" in Ullmann's Encyclopedia of Industrial Chemistry, 2007 Wiley-VCH, Weinheim. doi:x.1002/14356007.a03_325.pub2
- ^ Mendham, J.; Denney, R. C.; Barnes, J. D.; Thomas, M. J. Yard. (2000), Vogel'southward Quantitative Chemical Analysis (6th ed.), New York: Prentice Hall, ISBN0-582-22628-7
- ^ Meyer, K.; Bloch, H. S. (1945). "Naphthoresorcinol". Org. Synth. 25: 73; Coll. Vol. 3: 637.
- ^ Dark-brown, G. B. (1946). "Methylsuccinic acid". Org. Synth. 26: 54; Coll. Vol. three: 615.
- ^ Ford, Jared H. (1947). "β-Alanine". Org. Synth. 27: ane; Coll. Vol. 3: 34.
- ^ Anslow, W. Thousand.; King, H.; Orten, J. M.; Hill, R. One thousand. (1925). "Glycine". Org. Synth. 4: 31; Coll. Vol. i: 298.
- ^ Durham, Fifty. J.; McLeod, D. J.; Cason, J. (1958). "Methyl hydrogen hendecanedioate". Org. Synth. 38:55; Coll. Vol. 4:635.
- ^ Chaudhari, M. R.; Kulkarni, Y. A.; Gokhale, S. B. (6 October 2008). Biochemistry and Clinical Pathology. ISBN9788185790169.
- ^ Thorpe, J. F.; Kon, G. A. R. (1925). "Cyclopentanone". Org. Synth. 5: 37; Coll. Vol. 1: 192.
- ^ Conant, J. B.; Tuttle, Niel. (1921). "Diacetone alcohol". Org. Synth. 1: 45; Coll. Vol. one: 199.
- ^ Karabinos, J. V. (1956). "γ-lactone". Org. Synth. 36: 38; Coll. Vol. 4: 506.
- ^ Pradyot Patnaik. Handbook of Inorganic Chemicals. McGraw-Hill, 2002, ISBN 0-07-049439-8
- ^ "Endothermic Reactions of Hydrated Barium Hydroxide and Ammonium Chloride". UC San Diego. Retrieved two April 2014.
- ^ Endothermic Solid-Solid Reactions
- ^ a b Camp, Eric. "Endothermic Reaction". Univertist of Washington. Retrieved 2 Apr 2014.
- ^ "Endothermic solid-solid reactions" (PDF). Classic Chemistry Demonstrations. The Royal Society of Chemistry. Archived from the original (PDF) on 7 April 2014. Retrieved 2 April 2014.
External links [edit]
- Textile Safety Information Sheet (MSDS)
Ba Oh 2 Compound Name,
Source: https://en.wikipedia.org/wiki/Barium_hydroxide
Posted by: stonegared1969.blogspot.com






0 Response to "Ba Oh 2 Compound Name"
Post a Comment